Tariff system
Under the Act of 22 July 2014 amending the Act on publicly financed healthcare services and certain other acts, as of 1 January 2015, the Agency was assigned a new task of setting tariffs for healthcare services and preparing reports in this respect, as well as developing, verifying, collecting, making available and disseminating information on tariffs, and developing draft recommendations on the cost accounting standard applicable to healthcare providers.
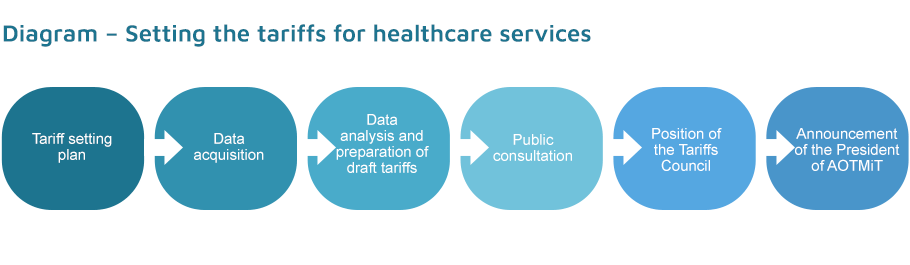
The tariff setting plan, each time drawn up for a given calendar year by 1 June of the preceding year and approved by the Minister of Health, is the basis for activities related to setting the tariffs for healthcare services. All tariff setting plans are published in the Public Information Bulletin.
The tariff setting plan (Tariff report) is based on service provider data obtained for all services in the healthcare system, and it is the result of analytical work on the data obtained during the procedures; the draft tariffs included in the report are subject to public consultations, and they are then reviewed by the Tariffs Council.
The President of AOTMiT decides on the tariff and publishes it by way of an Announcement of the President of AOTMiT in AOTMiT’s Public Information Bulletin, following prior approval by the Minister of Health.
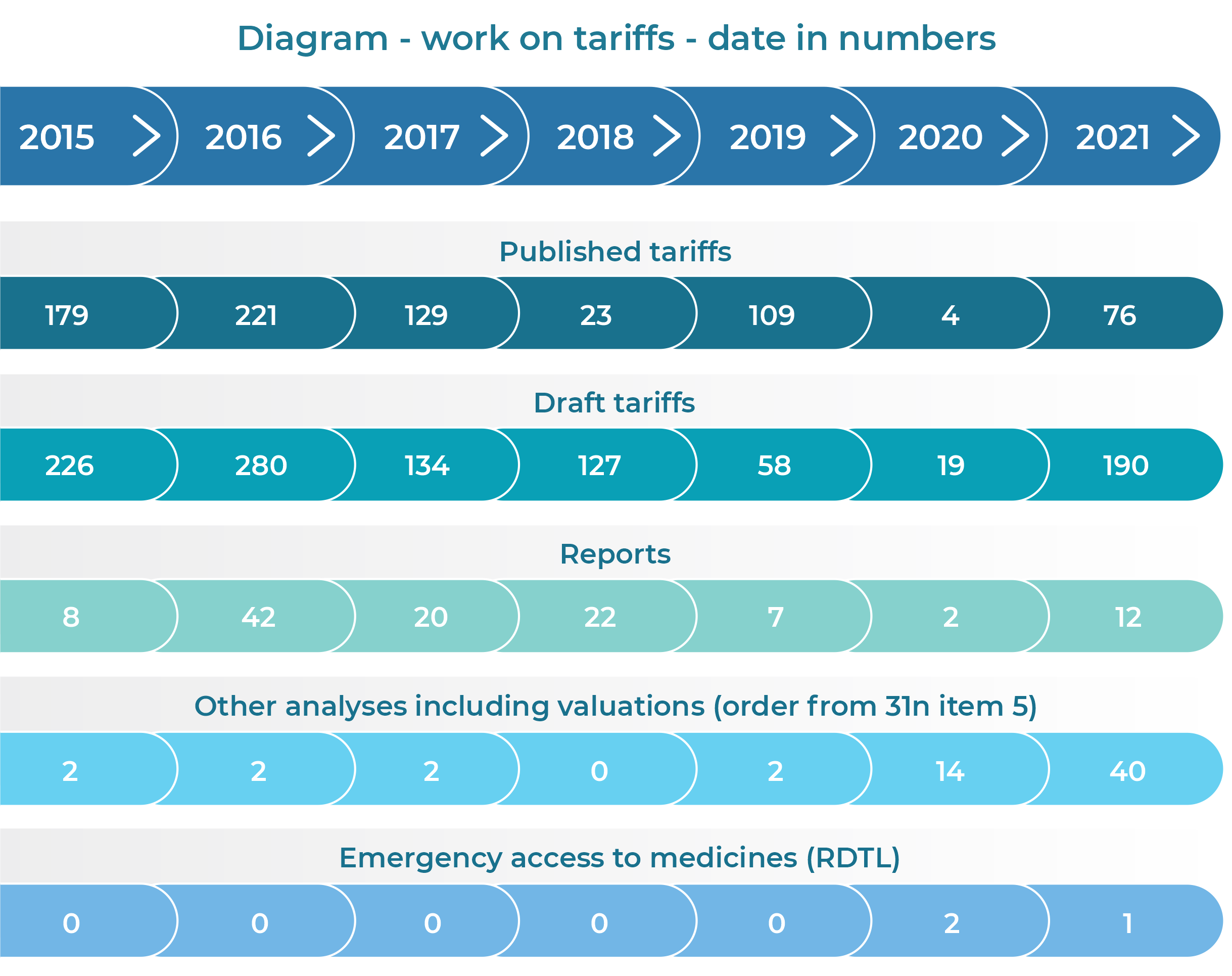
The Agency’s activities related to tariff setting and the cost accounting standard provide many benefits both to healthcare providers and to the entire healthcare system. Most notably, they provide information on the actual costs of the healthcare services provided, which ensures the proper distribution of funds by the payer. The purpose of the activities is to improve the system within the scope covered by the tariff.
*Ladies and Gentlemen
We inform you that the materials and results of the discussions posted on the Agency’s website are the result of the conceptual work and analytical process carried out by the team of the Agency for Health Technology Assessment and Tariff System based on the EBM paradigm, including: search, selection, synthesis and interpretation of scientific evidence, or the data analysis carried out.
In connection with the above, we would like to inform you that the use of analytical material or the results of the discussion, in accordance with good practice, should be accompanied by information on the source in the form: [title of presentation / report], AOTMiT, Warsaw, June 2024]

